ABSTRACT
This paper addresses how to qualify a Biological Safety Cabinet(BSC) and includes a description for Installation and Operational Tests that should be included as part of the Qualification process.
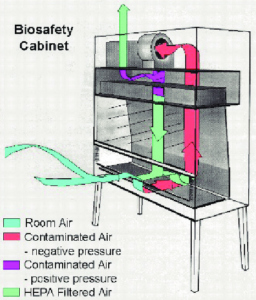
Despite having robust design and environmental monitoring, cleanrooms are at risk for several sources of contamination. In this case, BSCs, if purchased with much consideration, are the first line of defense in providing personnel, environmental, and/or product protection against biohazards and harmful agents in a range of laboratory, research, clinical, and pharmaceutical settings. As much as selecting the right BSC is important, it is more important to ensure that the BSC is functioning correctly at a high safety level. This white paper provides a template/ description of various Installation Qualification (IQ) and Operational Qualification (OQ) tests that should be included at a minimum when qualifying BSCs to ensure it helps facilitate an optimum working environment.