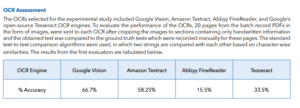
ABSTRACT
In drug manufacturing, keeping track of data is crucial for the drug’s approval from the FDA. The “Continued Process Verification” (CPV) data needs to be maintained for ensuring that the product outputs are within predetermined quality limits. In spite of rising demand for the creation of digital data directly at the source itself, some companies follow the traditional methods of documenting the processes parameters on paper, on designed forms. This leads to data being inaccessible for others unless it is again digitized by someone. The traditional way of achieving this is to have someone enter the data into a computer system by shuffling around the pages in the document. The manual process consumes a lot of time and leaves very little time for the data entered to be validated. This article briefly describes the possible methods of automating the manual data entry process, and how the upcoming technologies can be used for this work.